Breakthrough test can evaluate enzyme involved in process associated with cancer cell death
New research could pave the way to develop cancer drugs targeting an enzyme that inhibits ferroptosis, a type of cell death. Cancer cells that are resistant to anticancer drugs are known to be vulnerable to ferroptosis. Therefore, agents that effectively induce or enhance the sensitivity of cancer cells to ferroptosis are expected to become novel […]
New research could pave the way to develop cancer drugs targeting an enzyme that inhibits ferroptosis, a type of cell death. Cancer cells that are resistant to anticancer drugs are known to be vulnerable to ferroptosis. Therefore, agents that effectively induce or enhance the sensitivity of cancer cells to ferroptosis are expected to become novel anticancer agents.

Credit: Nakamura, T., and Ito, J., et al.
New research could pave the way to develop cancer drugs targeting an enzyme that inhibits ferroptosis, a type of cell death. Cancer cells that are resistant to anticancer drugs are known to be vulnerable to ferroptosis. Therefore, agents that effectively induce or enhance the sensitivity of cancer cells to ferroptosis are expected to become novel anticancer agents.
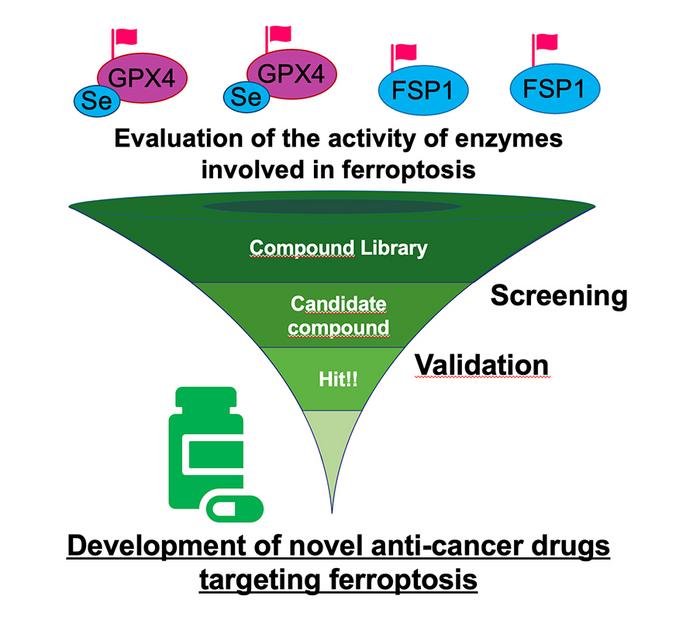
Research presented in a paper published in Cell Reports Methods on February 25 provides an appropriate and simple way to assess the enzymes essential to this process.
“Ferroptosis, a form of cell death, is noteworthy for its association with heightened sensitivity in cancer cells that are resistant to conventional anticancer treatments. This recognition underscores the pressing need for innovative therapies targeting ferroptosis,” said Junya Ito, a professor at the Laboratory of Food Function Analysis at Tohoku University. “In a collaboration with the research teams at Tohoku University and the Helmholtz Center Munich, a groundbreaking method has been developed to assess the enzymatic activity of glutathione peroxidase 4 (GPX4), a pivotal regulator of ferroptosis.”
Ferroptosis works through excessive lipid peroxidation, thereby destroying cells. The GPX4 enzyme can halt this process, allowing cells to avoid ferroptosis and cell death. However, monitoring for GPX4 activity can be challenging for multiple reasons, including because of the presence of other enzymes and proteins that can cause assays to over- or under-estimate the contribution of GPX4. In addition, GPX4 is a selenoprotein, which includes selenocysteine in its active site. This property makes it difficult to create recombinant proteins in bacteria as applied in conventional enzyme assays.
To overcome the challenges associated with evaluating GPX4 enzymatic activity, researchers developed a GPX4-specific assay that uses GPX4 collected from mammalian cells and purified lipid hydroperoxide. The straightforward approach presented in this paper not only accurately measures GLPX4 activity, but it also goes beyond the initial proposed application. “While conventional techniques have struggled to accurately measure GPX4 activity, this novel approach allows for precise evaluation by directly isolating the enzyme from cellular sources. Moreover, this method extends its utility beyond GPX4, offering a means to assess the activity of other enzymes involved in ferroptosis regulation,” said Ito.
The assay that researchers developed to identify GPX4 activity has many benefits. It uses standard equipment, is easy and convenient to develop, is versatile and can be used to measure other enzymatic activity, and it is scalable. In addition to developing an assay that adequately identifies GPX4 expression, it can also identify other enzymes that play a role in ferroptosis. For example, it can properly evaluate the effect of GPX4 inhibitors such as RSL3, which induces ferroptosis by blocking GPX4 function. It can also be used for FSP1, an enzyme that also inhibits ferroptosis. Using this method, researchers newly identified a compound having FSP1 inhibitory effect.
Looking ahead, researchers are optimistic about what this data means for the future of anticancer drugs. “Given its broad applicability, this innovative method promises to revolutionize the development and assessment of novel anticancer therapeutics targeting ferroptosis,” said Ito.
Other contributors include Toshitaka Nakamura, André Santos Dias Mourão, Adam Wahida, Eikan Mishima, and Marcus Conrad of Helmholtz Zentrum München; and Kiyotaka Nakagawa Tohoku University.
The Deutsche Forschungsgemeinscha, the Priority Program SPP 2306, the CRC TRR 353, the German Federal Ministry of Education and Research, and the European Research Council under the European Union’s Horizon 2020 Research and Innovation Programme, and the JSPS KAKENHI supported this research.
Journal
Cell Reports Methods
DOI
10.1016/j.crmeth.2024.100710
Article Title
A tangible method to assess native ferroptosis suppressor activity
Article Publication Date
25-Feb-2024
What's Your Reaction?

































